Application notes
Interparticle cohesion in metal powders - GranuHeap Analysis
The GranuHeap is an instrument able to estimate the cohesion of granular materials by comparing the shape of a true heap with the shape of a heap presumably ideal and free from cohesion.
Principle
The GranuHeap is an instrument able to estimate the cohesion of granular materials by comparing the shape of a true heap with the shape of a heap presumably ideal and free from cohesion. The obtained cohesive index authorizes a simple and fast comparison on various samples.
Therefore, several metal powders (granular materials recognized for the absence of surface interactions) were analyzed by our care. The principal characteristics of these powders diverge in their grain size, their shape and the nature of studied material.
LEARN MORE ABOUT THE GRANUHEAP
Description of the powders
Figures 1 and 2 present the particles characteristics of the studied powders.
Figure 1 shows the morphology of the particles and the figure 2 presents the grain size distributions expressed in volume and in number.

Figure 1: Electron micrographs of the metal powders
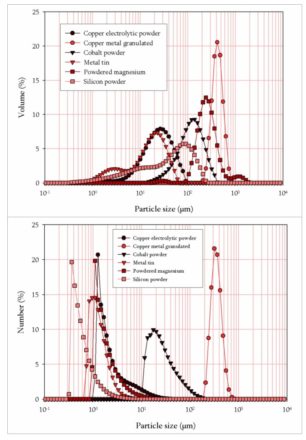
Figure 2: Grain size distributions expressed a) in volume and b) in number for a powder cohesion study comparison
The combination of these data shows
Copper metal granulated powder is mainly composed by spherical particles with size ranging between 200 and 800µm. The comparison of the distributions expressed in number and in volume (figure 2) indicates that this powder is relatively monodisperse.
Cobalt powder consists of pseudospheric compact grains with a lightly rough surface state. The size of the particles varies between 10 and 400µm.
Particles of the Powdered magnesium powder present a chips morphology with size varying between 100 and 1500µm (the numerical distribution is an artifact related to the measurement of the edges of the chips).
Electron micrograph of the Copper electrolytic powder shows that its particles have an atypical shape such as dendrites morphology. The grain size distribution expressed in volume extends from 1 to 100µm. The numeral distribution shows that the statistical weight of the small particles is significant (mode equal to 1.25µm).
Electron micrograph of the Metal tin powder shows that the grains are pseudospheric with relatively smooth quality surface. The grain size population seems to be relatively wide. This observation is confirmed by the grain size distributions.
On one hand, the figure 2a shows that the distribution varies from 700nm to 60µm and, on the other hand, the figure 2b illustrates the considerable weight carried by the particles ranging between 700nm and 4µm.
For the Silicon powder, grain size analysis expressed in volume shows that this powder consists of a double population with a total extent ranging between 700nm and 250µm. The distribution expressed in number varies between 300nm and 3µm. These results seem to agree with the observations carried out by scanning electron microscope where we can see (figure 1) simultaneously big angular massive particles accompanied by smaller.
LEARN MORE ABOUT THE GRANUHEAP
Results and Discussions
Table 1 shows the results obtained for the various metals. Three fields can be defined depending on the cohesive index value: a first lower than the unit, a second ranging between the unit and three and, finally, a last field defined above 10.
| Name of the powder | Heap Angle | Standard Deviation | Cohesive Index | Standard Deviation |
| Copper metal granulated | 30.1 | 1.5 | 0.6 | 0.3 |
| Cobalt powder | 38.7 | 1.1 | 0.1 | 0.0 |
| Powdered Magnesium | 40.5 | 0.4 | 0.3 | 0.1 |
| Copper electrolytic powder | 56.8 | 1.4 | 2.0 | 1.4 |
| Metal tin | 62.9 | 0.3 | 2.3 | 0.9 |
| Silicon powder | 64.2 | 3.6 | 10.9 | 2.1 |
Table 1: Heap angle and cohesive index of different metal powders in the case of a cohesion powder study comparison
As we will be able to see it, the division in fields comes from the particles characteristics (size and form) entering in the composition of the metal powders.
Field I: Cohesive index lower than the unit
It is not surprising that the Copper metal granulated powder presents the smallest slope angle of the series.
Indeed, the spherical shape of the particles and the monodisperse character of the grain size distribution are as many elements that confer to the powder a minimum of mechanical frictions supporting its good flow properties.
Because of a smaller average grains size (increasing the number of interparticle contacts), we can see that the slope angle of the Cobalt powder is slightly higher. The cohesive index conditioned by the pseudospheric shape of the particles remains lower than the unit. With others particles characteristics, the table 1 shows that the Powdered magnesium powder has however results comparable with those of the Cobalt powder.
An explanation of this may be related to the shape of the particles.
The chips shape, presenting a particular anisotropic geometry, gives to the particles the aptitude to be preferentially oriented according to their face and to improve, consequently, their relative superposition increasing, by this way, slightly the slope angle. Table 1 shows that the cohesive index of these three powders is lower than the unit.
This does not have anything astonishing; the materials studied having been deliberately selected for their absence of surface interaction.
Field II: Cohesive index ranging between 2 and 3
The results obtained for the Copper electrolytic powder and Metal tin show, on one hand, that the slope angle values become larger and, on the other hand, that this increase goes hand in hand with a light increase the cohesive index.
An explanation for this finds its origin in the shape particles for the Copper electrolytic powder sample or in the grain size for the Metal tin sample. Indeed, the electron micrograph of the Copper electrolytic powder revealed atypical particles with dendrites aspect.
This particular shape confers on the particles to favor tangles and to reduce the slipping. This state increases simultaneously the slope angle and the intergranular cohesion.
For the Metal Tin sample, it is not any more the shape of the particles (in this case, the morphology is pseudospheric) which increases the slope angle and the cohesive index but rather the presence of submicronic particles. These lasts increase considerably the interparticle contacts number raising, by this way, the global friction.
Field III: Cohesive index higher than 10
In the case of the Silicon powder, we can observe that the flow properties of the powder seem to be exclusively controlled by the submicronic particles (the grain size analysis shows that this powder is that which has the smallest particles of the series – see on figure 2b). Their presence increases significantly the interparticle contacts number raising, by the same way, the total cohesion of the medium.
LEARN MORE ABOUT THE GRANUHEAP
Conclusion
✓ These results show, on one hand, that the heap model used returns a very good estimate of the cohesive index related to the cohesion of the powders.
✓ On the other hand, they show that, in the case of metal powders of various origins, not only the shape of the particles modifies this cohesive index but also the numerical grain size distribution : as the proportion of submicronic particles increases, as the cohesive index increases.
✓ Finally note that to shorten the rheological properties of a powder to its mean value, its median value d0.5 or its morphology alone is not sufficient
✓ It is necessary at least to take account of its distributions (volume and number) and of the general morphology of the particles (free agglomerates, aggregates, particles).
✓ Indeed, the results obtained show that additional correlations must be considered.
